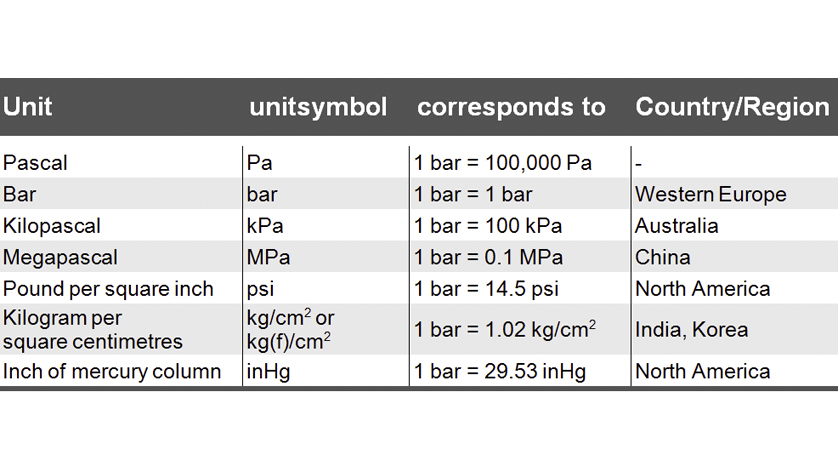
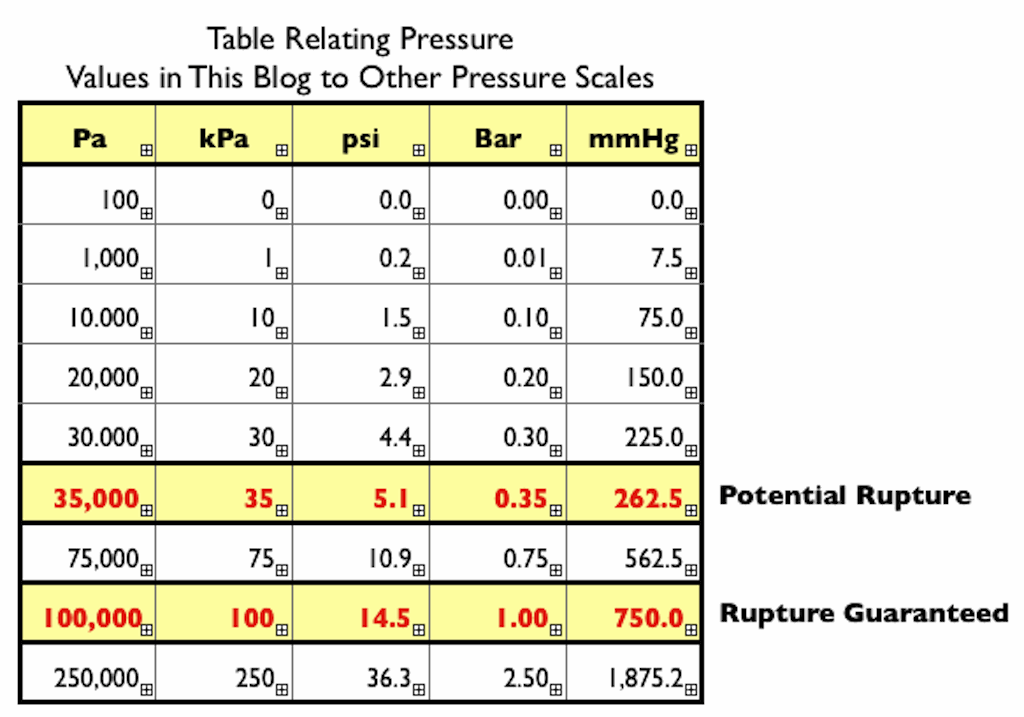
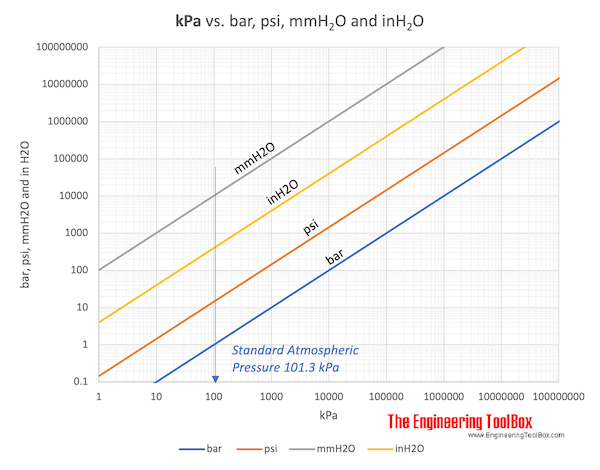
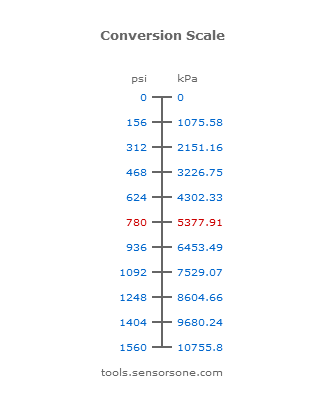
Atmospheres and Conversions: 1.00 atm = x 10 5 Pa = kPa = 760. Torr = 14.7 psi To convert pressure: 1.Turn what you have to atm 2.Multiply. - ppt download
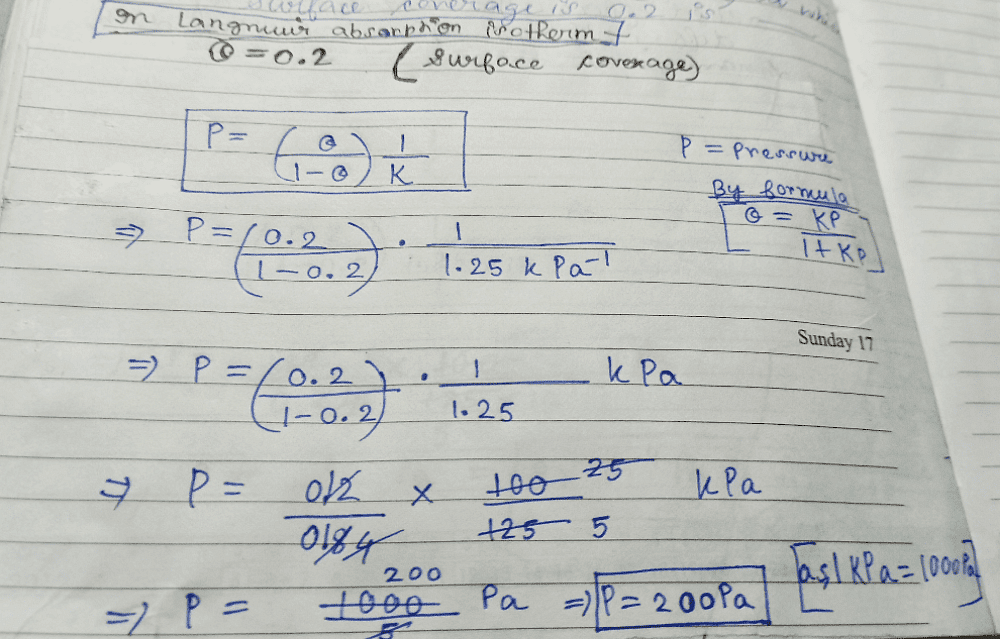
The adsorption of a gas follows the Langmuir isotherm with K = 1.25 kPa-1 at 25oC. The pressure in Pascal at which surface coverage is 0.2 is___? (rounded up to two decimal

Gas Laws Chapter 5. Pressure Force per unit area Measured in Atmospheres (atm) Mm of Hg = Torr Pascals or kiloPascals (Pa or kPa) - ppt download

SOLVED: quESt kPa? (1 atm 1.01325 measured be 654 mmHg What I5 the pressure The pressure of a gas sample Was 87.2 kPa 6,63 * 10*KPa 8.72 + 107 kPa 8,72 * 104KPa 118 KPa
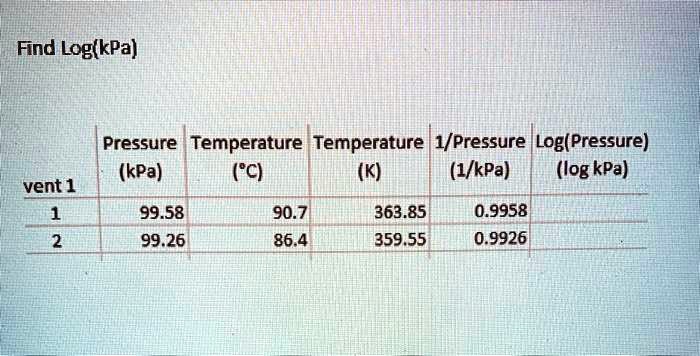
SOLVED: Find Log(kPa) Pressure Temperature Temperature | 1/Pressure |Log(Pressure) (kPa) (C) (K) (1/kPa) (log kPa) vent 1 99.58 90.7 363.85 0.9958 99.26 86.4 359.55 0.9926






![Bearing capacities [kPa] weightless soil, q s = 1 kPa | Download Scientific Diagram Bearing capacities [kPa] weightless soil, q s = 1 kPa | Download Scientific Diagram](https://www.researchgate.net/publication/336276044/figure/tbl4/AS:810609981550598@1570275888695/Bearing-capacities-kPa-weightless-soil-q-s-1-kPa.png)